Customer Background
The Santiago Longitudinal Study (SLS) at the University of Chile is a leading prospective birth cohort in Chile. For over 30 years, the NIH-funded study has followed participants born at a similar period of time (the cohort) from infancy into young adulthood. Originally focused on early-childhood nutrition, the study has evolved into an important platform for aging research, using multi-omics approaches to investigate how early-life exposures, such as obesity, may accelerate biological aging before clinical disease appears. Currently, the SLS has generated approximately 13,000 samples from study participants, which are contained in a biorepository housed at the Instituto de Nutrición y Tecnología de los Alimentos of Universidad de Chile and in Michigan, USA. They include plasma, serum, peripheral blood mononuclear cells (PBMCs), subcutaneous abdominal fat, and saliva, which are stored in 1.5 mL and 2 mL cryogenic vials. Each vial is preserved either in -80 °C freezers or liquid nitrogen to maintain sample integrity for long-term molecular analyses. Senior investigator is Dr. María Paulina Correa, epidemiologist specializing in chronic diseases. Management is carried out by María Ignacia García (BSc, Pontificia Universidad Católica de Chile), who oversees the collection, identification, and long-term storage of the biorepository’s biological samples.
The Sample Tracking Challenge
Maintaining accurate labeling and traceability is critical to protecting the scientific value of these specimens, particularly as the study expands into multi-omics research and international collaborations. Two years ago, García was tasked with modernizing the biorepository’s sample management system and implementing an online registry that allows investigators to locate specific samples more efficiently. During the reorganization of the sample inventory, two key challenges emerged. First, a percentage of older plasma, PBMC, and PBMC pellet samples, collected between 2021 and 2023, had been labeled directly onto cryovials using non-cryogenic markers. When these samples were removed from -80 °C storage, the handwritten identifiers began to smudge or bleed, making them difficult to read, and potentially compromising proper sample identification. While optimizing storage space within freezer boxes, García noticed that some labels were either fully detached or partially detached, causing them to get hooked on the tube dividers within the box and become further removed from the tube. The combined effects increased the possible risk of sample misidentification within the biorepository. García explains: “These samples represent decades of longitudinal research. Maintaining durable labeling and clear traceability under ultra-low temperature conditions was essential.”
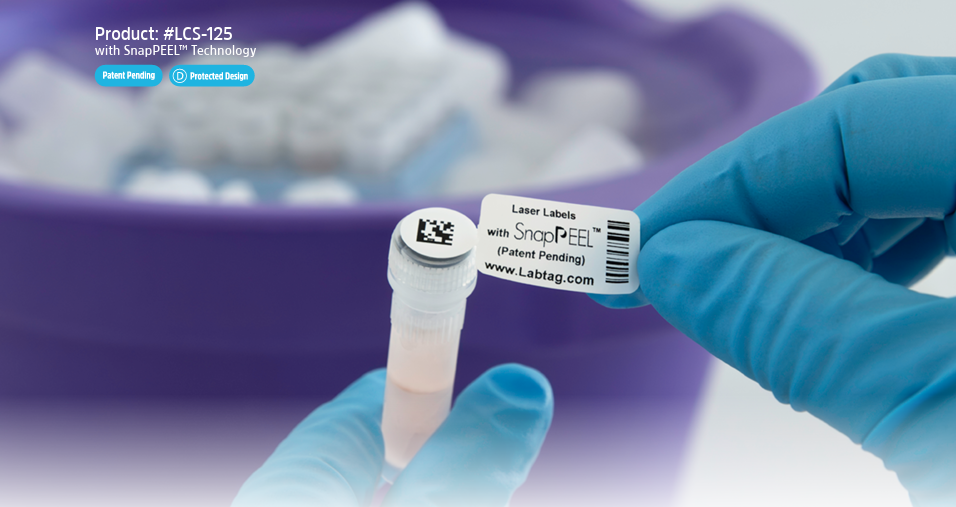
Tailoring a Sample Tracking Solution
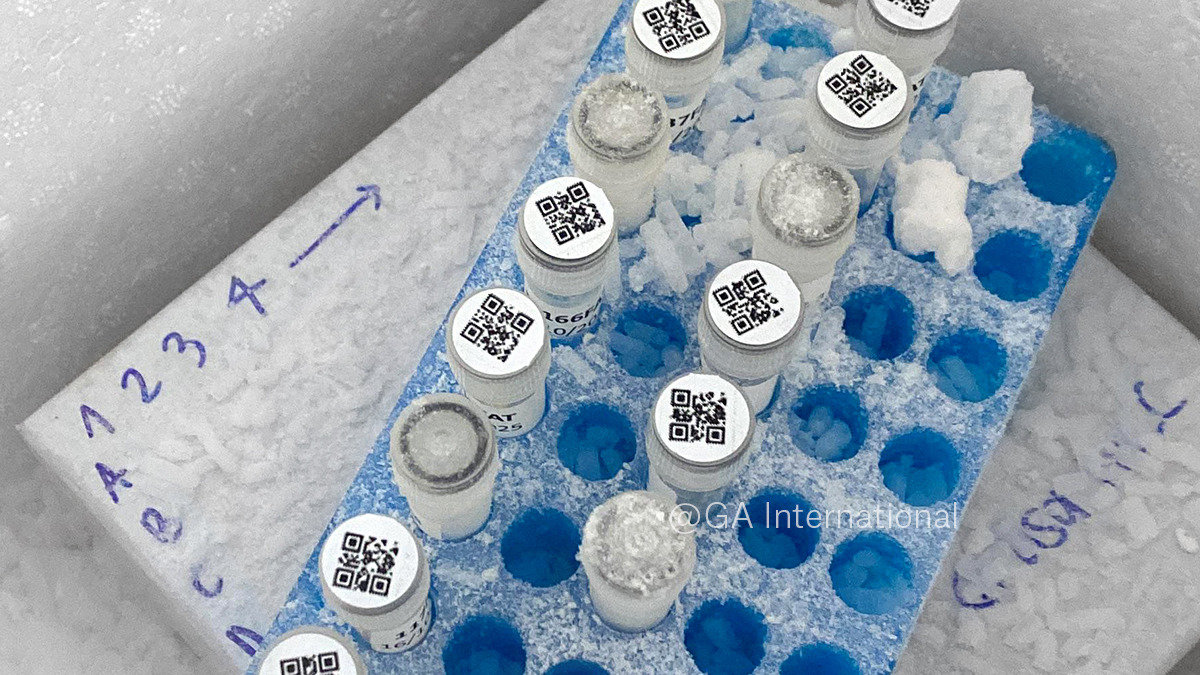
After discovering LabTAG, García requested free samples through GA International’s (LabTAG) website to support her relabeling efforts. She then connected with a LabTAG scientific expert, who gathered details about the biorepository’s workflows and labeling challenges. The solution that was recommended included LabTAG CryoSTUCK® labels. Specifically selected for their ability to be attached to surfaces at temperatures down to -80°C, and stored in liquid nitrogen, these labels were ideal for the necessary relabeling of the frozen biorepository samples. With guidance from LabTAG’s team, García standardized labeling practices across the biorepository. Each vial was assigned a unique ID number and clearly defined sample type, while storage boxes were reorganized using a universal structure to improve traceability and optimize space utilization. For collaborative projects involving outside laboratories, samples and aliquots are now prepared and labeled in advance to ensure consistent identification and faster processing for international shipments. Samples are sent with the required documentation, waivers, and permits to partner labs for specialized analyses, such as proteomics and epigenetic clock (biological age) studies. All collaborations to date have been successful, with samples consistently and correctly labeled. The processing of subcutaneous abdominal fat samples presented another challenge. Previously, these tissues were collected, placed inside prelabeled tubes, then flash frozen in liquid nitrogen before long-term storage at -80°C. Standard labels often lost adhesion following this freezing procedure, necessitating cumbersome relabeling and potentially risking sample integrity and misidentification.
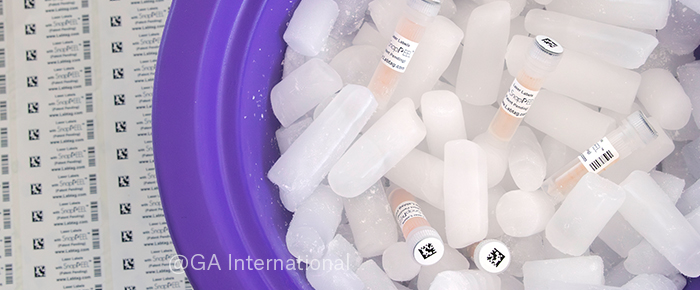
To address this, García transitioned these samples to CryoSTUCK® cryogenic labels, allowing cryovials to be re-labeled directly while frozen. The labels also contained SnapPEEL™ technology. Here, a rectangular and circular label are joined together, enabling dual identification of each tube, and allowing for removal of both sections from the label backing in one motion, saving time and effort. Taking advantage of CryoSTUCK®’s ability to stick to surfaces at -80 °C, while samples remained on dry ice, labels were applied quickly and securely. The main rectangular label, placed on the side of the tube, displayed the sample ID and processing date. The detachable circular section containing a QR code linked to the digital registry, was then easily detached from the rectangular label, and placed onto the tube cap, providing the additional verification layer. The more advanced QR code also allowed additional variables to be added, making sample tracking easier and more complete. Finally, LabTAG’s team also provided support in refining Word label templates, helping improve alignment and reduce printing errors. García remarked, “The labeling update of the SLS biorepository is still ongoing, with more work ahead. However, with the help of CryoSTUCK® labels, I estimate the process will be three times faster, as they adhere securely to the frozen surface of cryovials.”
Conclusion
This case study illustrates the value of implementing properly standardized cryogenic labels in a high-volume research biorepository such as the SLS. By replacing marker-based identification methods and poorly adhering labels with CryoSTUCK® cryogenic labels, García established a reliable labeling workflow. This system can withstand -80°C storage and liquid nitrogen exposure, while also complying with international sample handling standards. The result is consistent adhesion, dependable performance at ultra-low temperatures, and improved sample traceability, supporting both long-term aging research and global scientific collaboration.